
PHARMACEUTICAL SUPPLY
PARTNER
We strive to deliver on every promise and work extremely hard, so your facility and your patients have the lifesaving medications and supplies for every procedure and patient.

THE RIGHT WAY

We can help you locate whatever your practice requires on short notice, with minimal effort on your part, and at the most competitive price possible.

PHARMACEUTICAL SUPPLY
PARTNER
We strive to deliver on every promise and work extremely hard, so your facility and your patients have the lifesaving medications and supplies for every procedure and patient.

THE RIGHT WAY

We can help you locate whatever your practice requires on short notice, with minimal effort on your part, and at the most competitive price possible.
ABOUT US
MAKING SURE YOU HAVE WHAT YOU NEED TO SERVE YOUR PATIENTS
When you chose your career path, you did so from a firm belief that you can do good and help people regain their good health. Thus, it is beyond frustrating when forces out of your control prevent you from doing your job to your best abilities and resources.
Even before the COVID pandemic, supply disruptions have been an enormous obstacle for the healthcare facilities, their staff, pharmacists, doctors, nurses, and most importantly, their patients and their families. These ever so prevalent events compromise or delay important and often lifesaving drug therapies and procedures and significantly contribute to the rise in medication errors and adverse effects when alternatives have to be used.
No one company can fix this problem, but we will always do our absolute best. At Mercer Medical, our mission and purpose are to utilize all our resources and experience to ensure that you always have what you need to get the job done and at the lowest financial and emotional cost
Mercer Medical is a fully licensed and DCSA compliant pharmaceutical and medical supplies distribution company. We service private and government healthcare institutions, independent pharmacies, and doctor offices. We either stock or have access to most FDA-approved pharmaceutical and medical products.
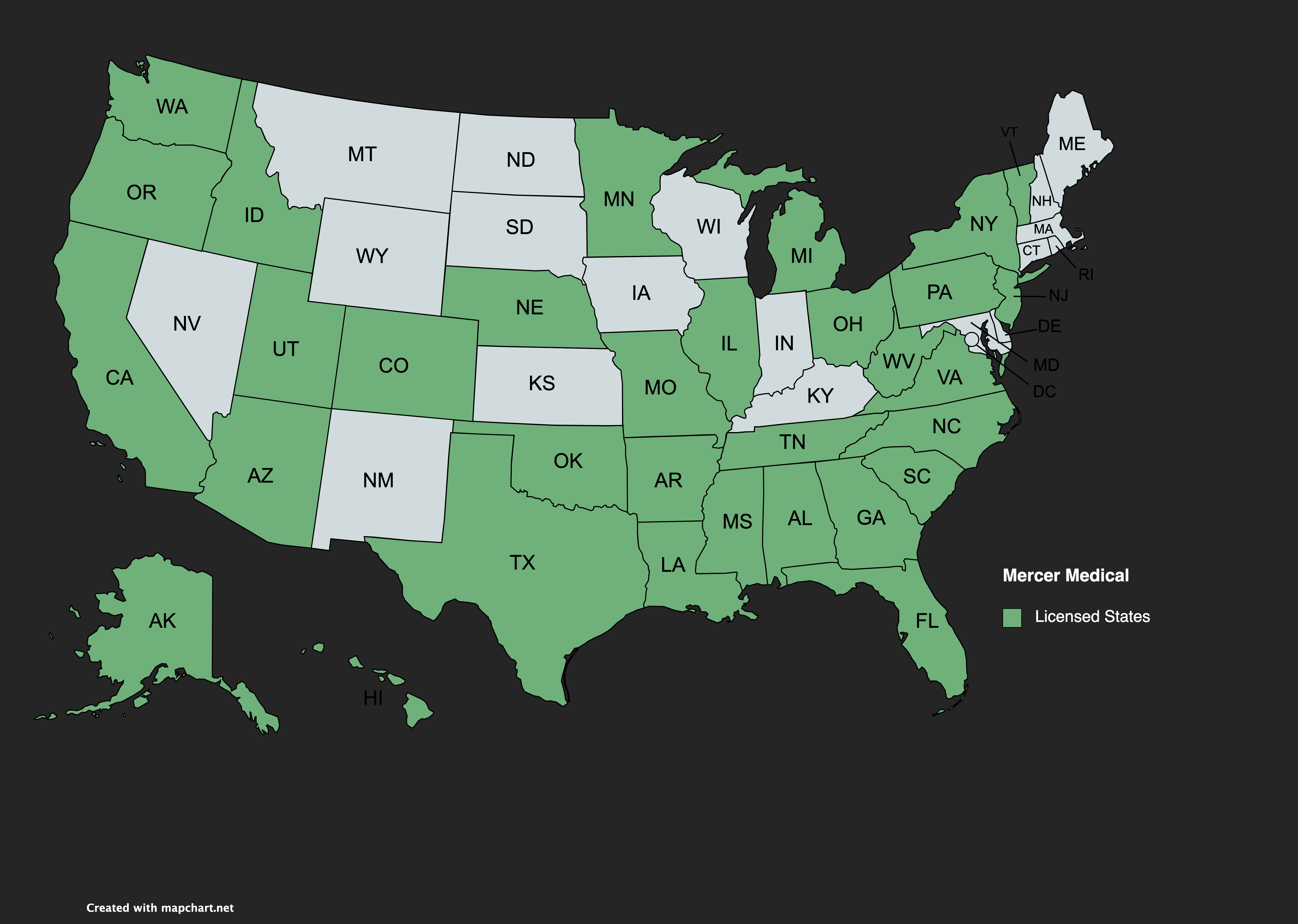
Our business was founded in Washington state in 2006, but we are currently operating in all major US states, including CA, FL, TX, and most East Coast states. Our company has a flawless record with all State and Federal regulatory agencies, and we are in full compliance with all provisions of DSCSA.
WE ARE LICENSED IN 30 STATES
ABOUT US
MAKING SURE YOU HAVE WHAT YOU NEED TO SERVE YOUR PATIENTS
When you chose your career path, you did so from a firm belief that you can do good and help people regain their good health. Thus, it is beyond frustrating when forces out of your control prevent you from doing your job to your best abilities and resources.
Even before the COVID pandemic, supply disruptions have been an enormous obstacle for the healthcare facilities, their staff, pharmacists, doctors, nurses, and most importantly, their patients and their families. These ever so prevalent events compromise or delay important and often lifesaving drug therapies and procedures and significantly contribute to the rise in medication errors and adverse effects when alternatives have to be used.
No one company can fix this problem, but we will always do our absolute best. At Mercer Medical, our mission and purpose are to utilize all our resources and experience to ensure that you always have what you need to get the job done and at the lowest financial and emotional cost
Mercer Medical is a fully licensed and DCSA compliant pharmaceutical and medical supplies distribution company. We service private and government healthcare institutions, independent pharmacies, and doctor offices. We either stock or have access to most FDA-approved pharmaceutical and medical products.
Our business was founded in Washington state in 2006, but we are currently operating in all major US states, including CA, FL, TX, and most East Coast states. Our company has a flawless record with all State and Federal regulatory agencies, and we are in full compliance with all provisions of DSCSA.
WE ARE LICENSED IN 30 STATES

PRODUCTS & SERVICES
Work with a Distributor Who Delivers Every Order
on Time!
We can supply any non-controlled pharmaceutical or medical supply product used by a hospital, surgery center, doctor's office, or an independent pharmacy.
ORAL MEDS
INJECTABLES
SOLUTIONS
VACCINES
DME
PPE
CLEANING SUPPLIES
SURGICAL SUPPLIES
